

|
Orion Nebula: the red light is (mostly) hydrogen Balmer series |

Jakob stepped back to admire what he had written last: the concise equations of an ideal mass point moving under the influence of elastic forces. How beautiful classical physics is! Night Thoughts of a Classical Physicist McCormach
Jakob stepped back to admire what he had written last: the concise equations of an ideal mass point moving under the influence of elastic forces. How beautiful classical physics is! (Was! He meant before the Quantum Theory) Night Thoughts of a Classical Physicist McCormach
The layer of prejudices we acquire before we are sixteen" A. Einstein
| Found black lines superimposed on sun's spectrum. This is the sun's spectrum "folded up" |  |
| Heated gases give characteristic wavelengths, and can match dark lines in solar spectrum to bright emission lines: e.g. Sodium | 
|
| Hydrogen is simplest | 
|

| Light falls on metal, ejects negative charge, which we now know consists of electrons: can measure energy of electrons by stopping them in an electric field | 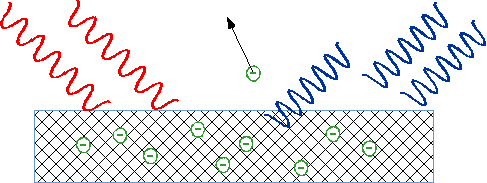 |
| No problem, since light carries energy? |  |
| (Thomson) proposed currant bun model of atom,: electrons imbedded in positively charged material. |  |
T(K) = T(oC) + 273
| To derive a theory of Black body radiation put E.M. standing waves in cavity. no particular reason to assume that any one wavelength preferred to any other: short waves dominate (since there are more of them) |  |
| this gives Rayleigh-Jeans curve. The "ultra-violet catastrophe": all radiation should be very short λ. and should not depend on temp. Wien noticed that experimental curve looked like Maxwell-Boltzmann distribution. for molecules |  |
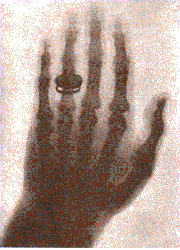
| Röntgen (1895) |  |
| Very penetrating radiation produced by vacuum tube (requires pot. diff ~ 20 kV), which passes through solids, fogs photographic plates | 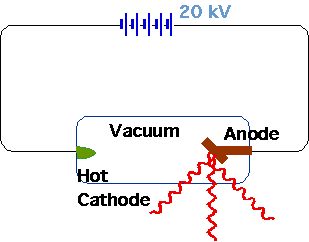 |
 |
 |
| Lead block with radium salt: α-particles are produced by radium, collimated by screen to narrow beam, pass through gold foil and are detected by scintillator (produces spark of light when hit by charged particle | 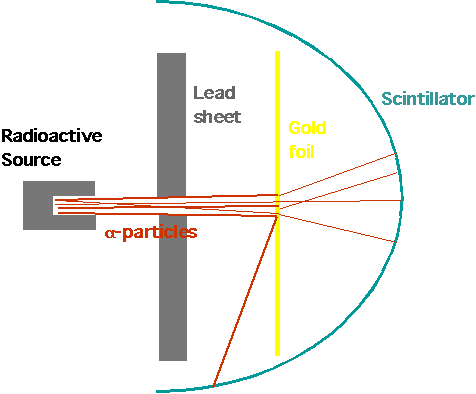
|
| Expect: rather small angle of deflection due to many small scatters | 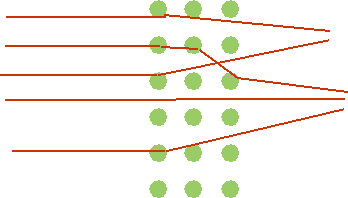
|
Close encounter with electron?
me ~ 1 mα 8000 |
 |
|
Cannonball bouncing backwards off tissue paper. Rutherford Need small, massive, positive charged nucleus. Electrons irrelevant! |
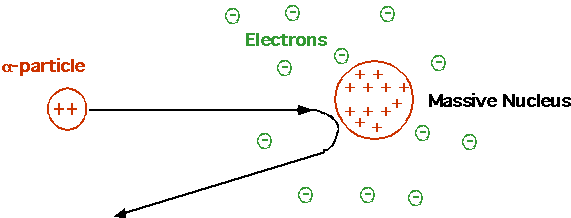 |
| Why don't the electrons fall into nucleus? |  |
| Maybe electrons are in orbit but an accelerating charge emits radiation so orbiting electrons should emit radiations and lose energy. |  |
E = 1/2 m v²
f λ = c
E = h f
h = 6.6 x 10-34 Js = 4.1 x 10-15 eV s
Since particles have energy, photons must carry energy (obviously light has energy: that's how you get a sun tan!)
| Einstein (1903): (note this was what he actually got the Nobel prize for) adapted Planck's hypothesis to say that light is absorbed in quanta (photons) most energetic electrons in metal need extra boost to escape: |  |
K.E. = 1/2mv²= hf-φwhere φ is work function: differs for different metals. Slope is universal ⇒ Planck's constant |
 |
| Electrons are ejected as soon as light strikes (no need for energy to accumulate), since photons arrive randomly | 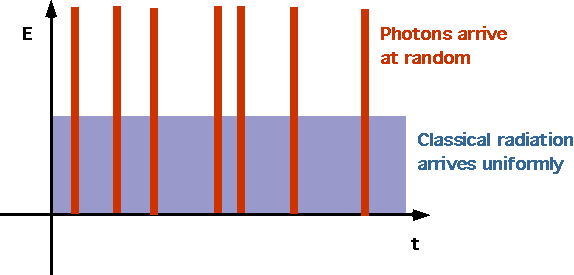 |
| "Classical" corks bob up and down | 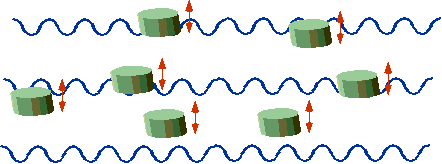 |
| "Quantum" corks are either stationary or ejected. | 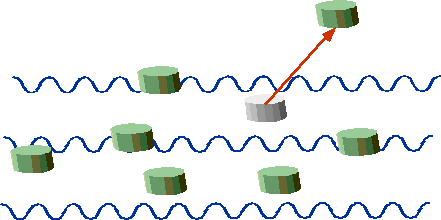 |
Kerner: Look at the edge if the shadow. It is straight like the edge of the wall that makes it. This means light is ..little bullets. Bullets go straight.
Hapggod (Tom Stoppard)
Kerner: When you shine a light through two little gaps, side by side, you don't get particle patterns like for bullets, you get wave patterns like for water. The two beams of light mix together
Hapggod (Tom Stoppard)
| Note that the shorter the wavelength, the more like a particle, so γ-rays (produced in nuclei) are usually treated only as particles. | 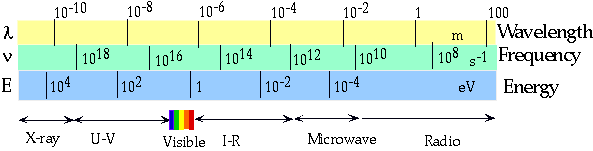 |
Model for H. atom must explain
| used crystal as diffraction grating: (2-D so pattern is more complicated) |  |
| A simpler experiment is now possible: the electron analog of Young's slits. Very low energy electrons pass through slits and hit detector (e.g. photo plate) and give 2-slit interference pattern | 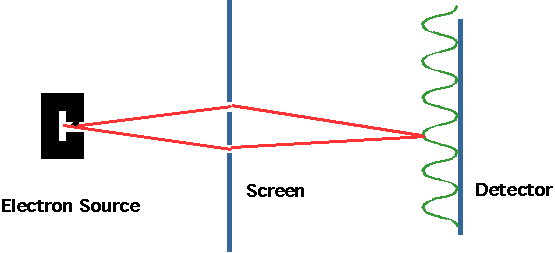 |
| you can even watch how it builds up |
| A dramatic recent example uses a buckyball C60 American Journal of Physics, Vol. 71, No. 4, p319, April 2003, Nairz, Arndt, and Zeilinger |
 |
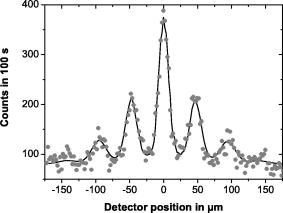
| Apparatus uses a diffraction grating:velocity v = 117 ms-1 |  |
| Circles are the experimental data. Line represent the model
A buckyball C60 has a λ ∼ 10-11m, but its "size" is 100 times bigger (∼ 10-11m) |
 |
| Newton:
\color{red}{
E = \frac{1}{2}mv^2 }
De Broglie:
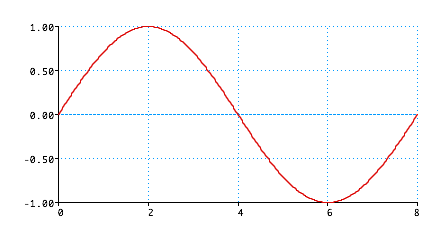
Standing wave
\color{red}{
\lambda = \frac{h}{p}}
Wave (like guitar string)
\color{red}{
\lambda = \frac{{2L}}{n}}
|
|

Model for H. atom must explain
n = 1 m = 2, 3, 4... n = 2 m = 3, 4, 5... n = 3 m = 4, 5, 6...
| De Broglie suggested that allowed orbits have an integral number of waves fitted into one orbit
\color{red}{
\lambda = \frac{{2\pi r}}{n} = \frac{h}{p}}
|
 |
because!
| These levels have energies
\color{red}{
E_n = -\frac{{13.6}}{{n^2 }}eV}
|
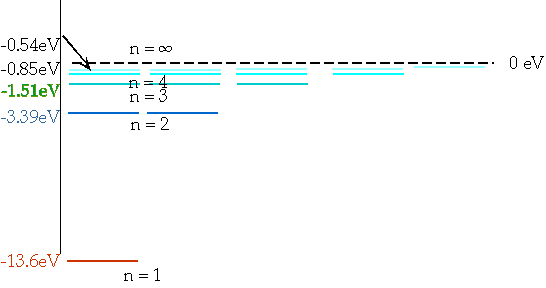 |
En = = - 13.6 eV
n²
e.g. n = 3 ⇒ n = 2δE = E3 - E₂ = 1.9 eV |
 |
|
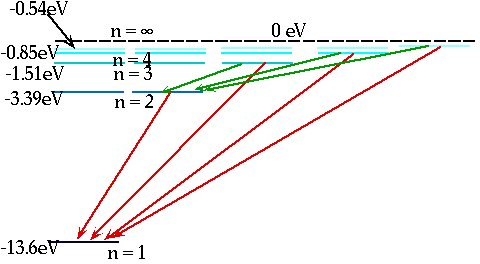 |
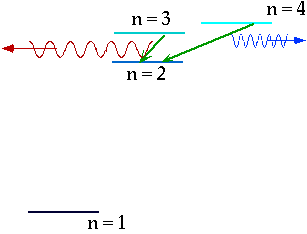
| If electron makes transition from one level to another, we will get emission line of definite energy | 
|
| However, if we have photons of all energies, one may have exactly the energy to raise the energy of an electron | 
|
| With care, can see both absorption and emission at the same time. | 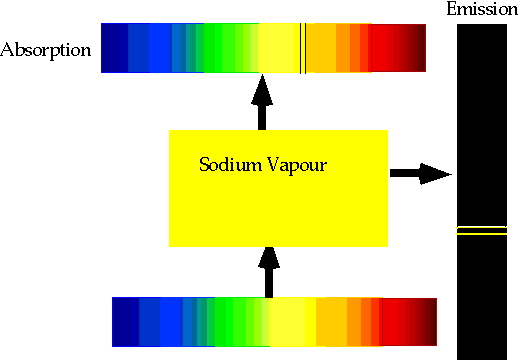 |
| Electron accelerated |  |
| Electron collides with atom, knocks out electron in lowest energy level, leaves vacancy for electron in higher level to fall into.e.g. for e.g Chromium: (Z = 24) | 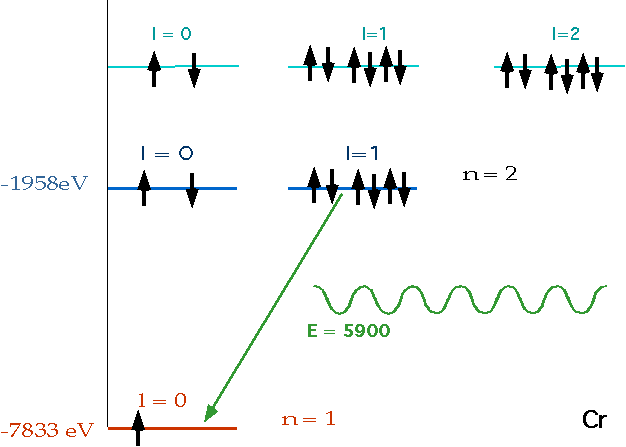 |
such as Giant Magneto_Resistance (GMR) Remember?